
This test was selected for the field study because it was one of the first Ag-RDTs to be granted US Food and Drug Administration Emergency Use Authorization and is widely in use in the US and internationally. Īs part of a COVID-19 household-transmission field study in Southern California initiated in November 2021, participants performed a daily at-home nasal swab Ag-RDT (Quidel QuickVue At-Home OTC COVID-19 Test) and self-collected saliva, anterior nares swab, and oropharyngeal swabs for reverse-transcription quantitative polymerase chain reaction (RT-qPCR) testing. 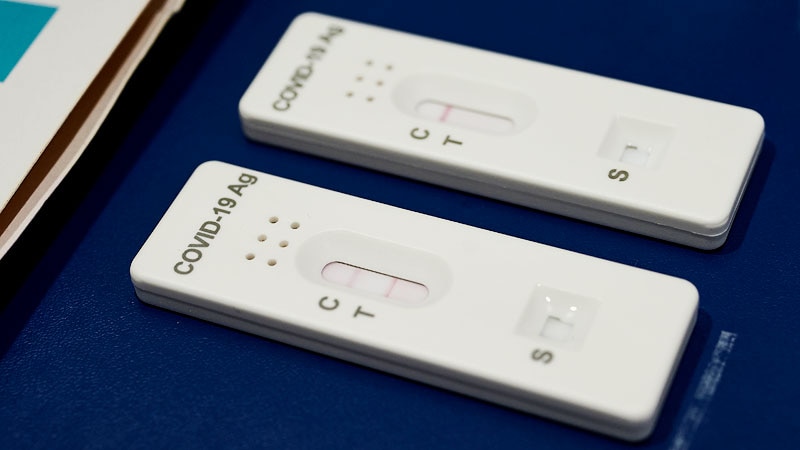
However, with widespread use imperfect specificity can result in many false-positive results, and at low prevalence of infection, these false positives can represent a large fraction of or even dominate among all positive results. Īg-RDTs typically have very high specificity of the 51 Ag-RDTs currently authorized for at-home use in the US as of 2 September 2022, all are required to demonstrate false-positive rates of ≤2%. Ag-RDTs are also used widely for test-to-enter events and serial screening testing in schools and workplaces for example, in May 2022, the California Department of Public Health began recommending Ag-RDTs as the primary test for COVID-19 in schools. Usage of at-home Ag-RDTs in the United States (US) has increased nearly 4-fold among those with self-reported coronavirus disease 2019 (COVID-19)–like illness between the period of Delta (23 August–11 December 2021) to Omicron (19 December 2021–12 March 2022) variant predominance. FDA's requirements for EUA approval.Antigen rapid diagnostic tests (Ag-RDTs) are increasingly used for detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). For example, the New York Times reported that Quidel’s antigen test by the University of Arizona detected only 32% of infections in asymptomatic individuals, despite the test meeting the U.S. While newer tests have aimed to positively identify active cases of COVID-19 in at least 80-90% of cases, and preferably higher, there is wide variance between antigen tests currently available on the market and the accuracy depends on the context of use. Generally, concerns about antigen tests are usually around its rate of false negatives, but recent outbreaks and articles have concerned many in public health and medicine about rates of false positive and false negative results. As more of these tests are produced and used on a wide scale, we hope to learn more about their accuracy and achieve as sensitive (correctly identifying those who are are actively infected with the virus) and specific (correctly identifying those who do not have an active infection) as possible. FDA has granted Emergency Use Authorizations (EUAs) for a handful of the more accurate antigen tests that are available. 
For example, as of early December 2020, the U.S. Several countries have begun authorizing the use of newer antigen tests that report lower rates of false positives and false negatives. While antigen tests can usually diagnose active COVID-19 infections, they are more likely to miss an active infection in comparison to molecular tests like polymerase chain reaction (PCR) tests. Several experts recommend using a second test to confirm a patient is truly negative or positive, particularly when patients may have no symptoms or have not been exposed to people who tested positive for the virus. 
While most newer antigen tests aim to accurately identify people with active COVID-19 infections at least 80% and 90% of the time (true positive rate), some antigen tests have been reported to have false positive or false negative rates as high as 50%. These considerations have a large impact as their effects can directly result in health impacts for people who test positive (but are not) and are quarantined with people with active infections or receive treatments like medication when it may be harmful. Food and Drug Administration (FDA).ĭespite having low rates of false positives, these types or errors in antigen tests still exist due to technical issues like handling, contamination, or test errors. Comparatively, false positive test results, which incorrectly show that a healthy person is infected by the virus when they are not, are very rare in tests that have been approved by regulatory agencies like the U.S. However, one of the major downsides of these tests is their high rate of false negative results (having a negative test result even if you are actively infected with the virus). Antigen tests for COVID-19 have many advantages, including rapid results, cheap production costs, and a high rate of accurate test results for people who are actively infected with COVID-19.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
